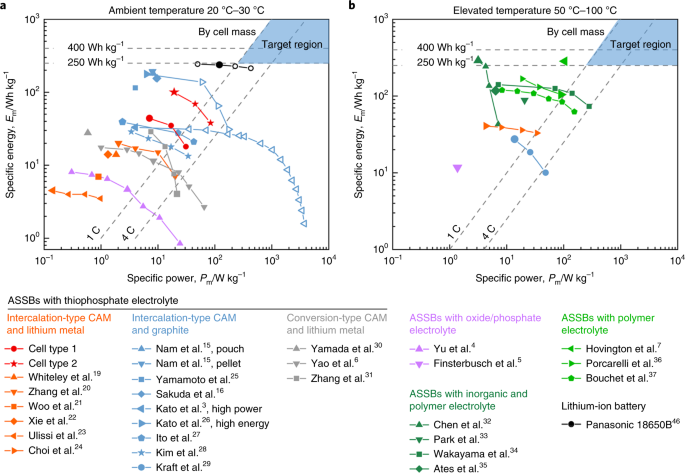
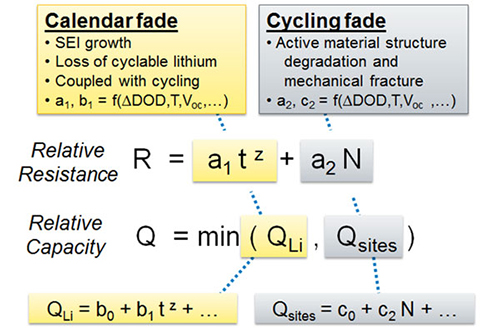
High Coulombic efficiency aluminum-ion battery using an AlCl3-urea ionic liquid analog electrolyte | PNAS
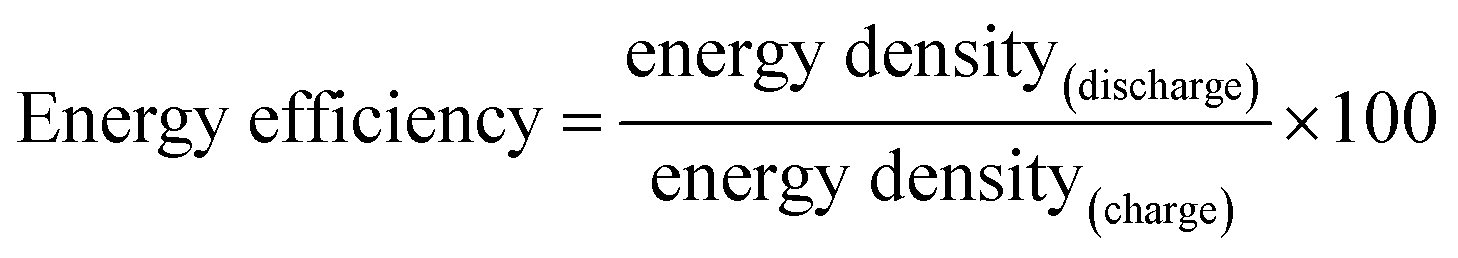
Energy efficiency: a critically important but neglected factor in battery research - Sustainable Energy & Fuels (RSC Publishing) DOI:10.1039/C7SE00350A

Battery energy storage efficiency calculation including auxiliary losses: Technology comparison and operating strategies | Semantic Scholar

SOLVED:Calculate the theoretical specific energy of the aluminum-air battery. The two electrode reactions are Al + 3OH; 6 AI(OHJ: 3e (-2.31 V) 02 2HzO + 4e 6 4OH (0.401 V)
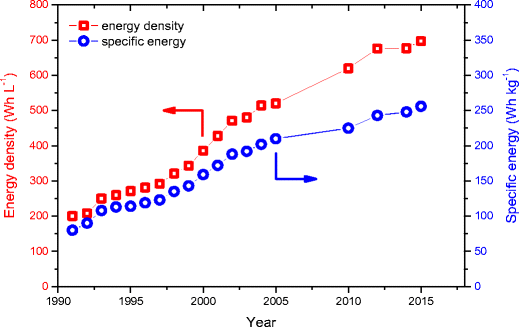
Lithium ion, lithium metal, and alternative rechargeable battery technologies: the odyssey for high energy density | SpringerLink

Metal–Air Batteries with High Energy Density: Li–Air versus Zn–Air - Lee - 2011 - Advanced Energy Materials - Wiley Online Library



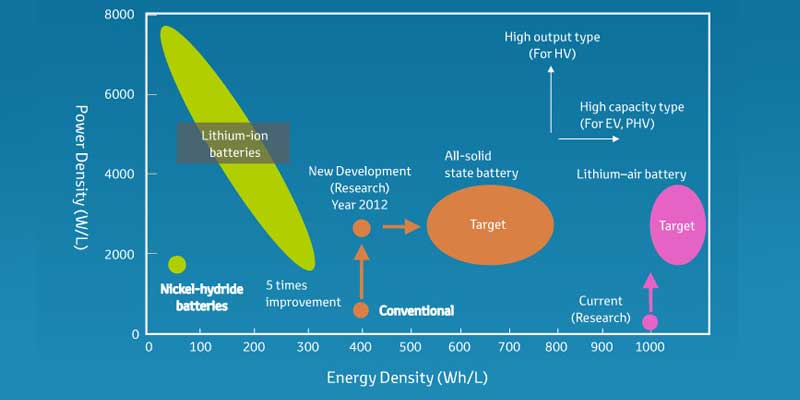

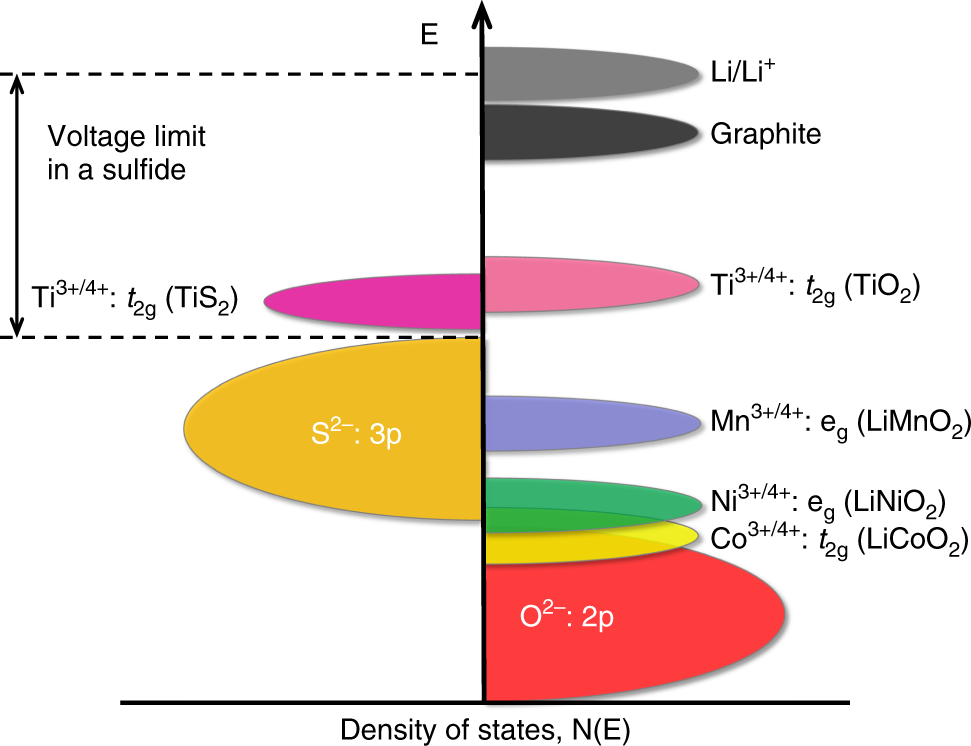



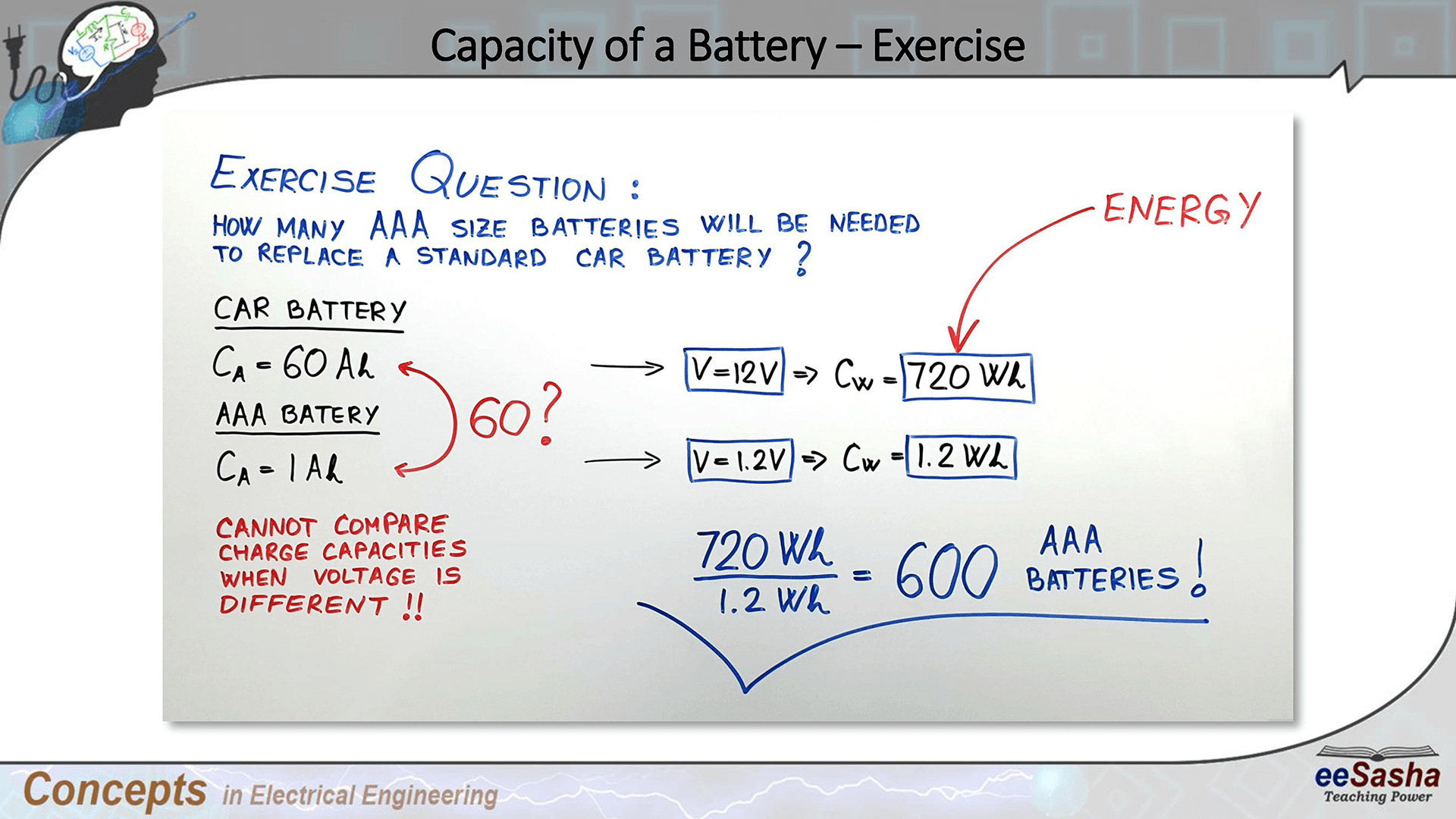
![Specific power and specific energy of di ff erent battery types [3]. | Download Scientific Diagram Specific power and specific energy of di ff erent battery types [3]. | Download Scientific Diagram](https://www.researchgate.net/profile/Daniel-Mueller-29/publication/257984459/figure/fig1/AS:297680254324736@1447983911716/Specific-power-and-specific-energy-of-di-ff-erent-battery-types-3.png)